StemEnhance Ultra: The World's #1
Stem Cell Nutrition Supplement,
Supports Your Body's Natural Release of
Bone Marrow Stem Cells...
StemEnhance Ultra:
Ultimate Stem Cell Support
StemEnhance Ultra is the result of 18 years of research and is the most effective and scientifically proven stem cell nutrition product on the market today.Through multiple clinical trials, StemEnhance Ultra was documented to optimize stem cell function in the body by increasing the number of both stem cells and Endothelial Progenitor Cells (EPCs) in the blood circulation, supporting optimum renewal and repair of tissues and organs.
StemEnhance Ultra concentrates and combines extracts from nature's most primitive superfoods, fresh water microalgae and marine macroalgae, providing the body with the ultimate in stem cell nutrition support.
New supplement formulation with the original StemEnhance, extracted from Lake Klamath harvested, blue-green algae (Aphanizomenon Flos-Aquae).

StemEnhance Ultra: Ingredients
StemEnhance Ultra concentrates and combines extracts from nature's most primitive superfoods, freshwater microalgae and marine macroalgae, providing the body with the ultimate in stem cell support.StemEnhance Ultra is backed by years of identifying, refining, and extracting compounds from blue-green algae using proprietary and patented technologies.
The blending of StemEnhance Ultra with Fucoidan offers a unique synergy that is further enhanced by Mesenkine - a unique extract from Spirulina isolated through our patented extraction process.
Mesenkine was shown to increase the blood concentration of G-CSF that plays a role in the release of adult stem cells. Mesenkine also supports the migration and homing of stem cells by balancing key messengers involved in stem cell function.
Through multiple clinical trials StemEnhance Ultra was documented to optimize stem cell function in the body by increasing the number of both CD34+ stem cells and Endothelial Progenitor Cells (EPCs) in the blood circulation, supporting optimum renewal and repair of tissues and organs the undergo breakdown and renewal as part of the normal aging process.
StemEnhance Ultra assists the body's inherent ability for long-term self-renewal by supporting the body's natural release of bone marrow stem cells.
StemEnhance Ultra is the result of 18 years of research and constitutes the most efficacious and scientifically proven stem cell support product on the market today.
We need vitamins, minerals and antioxidants to nourish our cells and we need alkaline water to flush away toxins. But none of this rebuilds the body. None of this can actually bring back new tissue, bone, organs, or any part of the body to support optimal health.
Science has just recently discovered that the only system known to actually rebuild the body is your own adult stem cells. If they are not releasing into the bloodstream as they should, your health will suffer.
It's as simple as that.
In Chinese medicine, Jing is of the utmost importance. Our congenital energy, the stronger our Jing, the longer and healthier we live. Bone marrow stem cells (when released into the blood stream) repair our body on a daily basis. And according to the Chinese, Jing is stored in the bone marrow. As we age, optimum health decreases as Jing decreases.
Renewing the body helps to balance hormones, and keep all internal systems functioning efficiently -- the way they should be.

StemEnhance Ultra: Proven Clinical Studies
Through multiple clinical trials, StemEnhance Ultra was documented to optimize stem cell function in the body by increasing the number of both stem cells and Endothelial Progenitor Cells (EPCs) in the blood circulation, supporting optimum renewal and repair of tissues and organs.
Placebo did not show any significant effect on the number of circulating stem cells, StemEnhance triggered a transient increase in the number of CD34++ cells (25%) and a long-lasting increase in the number of circulating EPC (>75%).
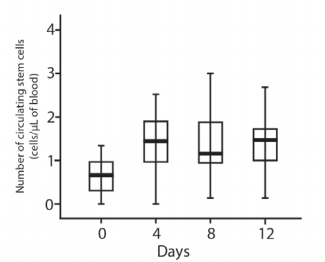
Consumption of Fucoidan from Undaria Pinnatifida led to an increase in the background level of circulating stem cells.
Stroke treatment and Endogenous Stem Cell Mobilization. Taken from: The Therapeutic Potential of Stimulating Endogenous Stem Cell Mobilization
Mobilization of human stem cells study published in the Journal of Cardiovascular Revascularization Medicine
StemEnhance does not promote tumor growth study published in AntiCancer Research Journal
Understanding the Natural Role of Stem Cells in the Body: A New Understanding of Disease Formation? Published in the Journal of Stem Cell Research & Therapy
Fucoidan ingestion increases the expression of CXCR4 on human CD34+ cells. Published in Experimental Hematology.
Consequences of AphanizomenonFlos-aquae(AFA) extract on metabolic profile of patients with type 2 diabetes published in Journal of Diabetes & Metabolic Disorders.
Mobilization of bone marrow stem cells with StemEnhance improves muscle regeneration in cardiotoxin-induced muscle injury published in Cell Cycle.
The Therapeutic Potential of Stimulating Endogenous Stem Cell Mobilization published in the Journal of Tissue Regeneration - From Basic Biology to Clinical Application.
Use of Stem Cell Mobilizer on Ankle Injuries to Expedite Recovery in Professional
Soccer Players published in MOJ Cell Science & Report.
Fucoidan protects mesenchymal stem cells against oxidative stress and enhances vascular regeneration published in International Journal of Cardiology.
Therapies from Fucoidan; Multifunctional Marine Polymers published in Marine Drugs.
StemEnhance Ultra also contains Mesenkine that was shown to increase the blood concentration of G-CSF that plays a key role in stem cell release.

StemEnhance Ultra Testimonials & Reviews
 Beth & Bill
Beth & Bill"StemEnhance Ultra has worked for both Beth and I and we will be taking Cerule products everyday for the rest of our lives.
Boosting stem cell release seemed to me to be the best type of health restoring supplement I could possibly take."
Cindy Olsen, Washington State
"StemEnhance Ultra is the best ever! Coupled with PlasmaFlo, I have never felt better. My mood is uplifted and energy has improved, my digestive system works great, my knee that I had surgery on has finally healed and my skin everywhere is slowly improving."
Mark A, Australia
"My hair grew back thicker than before so I feel much more like the younger me!"
 Frank Condon, World Class Runner
Frank Condon, World Class Runner"With StemEnhance Ultra, my energy level and recovery rate is so much better than that of other men in my age group. One guy who has known me since college approached me after watching my latest race and said, 'After seeing you today, I need to try whatever you are taking!'
So you can see that sharing StemEnhance Ultra with people is easy. When I can dominate in my age group (and usually beat younger competitors' times, too), I am a 'running advertisement' for StemEnhance Ultra and all Cerule's nutritionals!"
Sandra B, Australia
"I have been on StemEnhance Ultra and PlasmaFlo for nearly 3 years and my general health is so much better. I feel I am getting younger each year. People have commented that I look better and I certainly feel better. I believe that these are the best anti-aging products you can take."
 Yvonne F, FL
Yvonne F, FL"At 74, I am experiencing even more energy and stamina with StemEnhance Ultra.
I feel happier and my memory has improved as well.
I am a huge fan of StemEnhance Ultra!"
Mark P, Boston, MA
"Our whole family takes StemEnhance Ultra, and we all agree that it is a phenomenal product. We have more energy and focus - both which last throughout the day. And I have noticed that the frequent discomfort I used to feel with my off-and-on running routine has disappeared since StemEnhance Ultra became part of my daily regimen. This is one product that no one can do without!"
 Linda H, Australia
Linda H, Australia"I have been taking StemEnhance Ultra for 5 years and my health just gets better every year.
I feel my foundation health is stronger."
Marco A
"My active participation in soccer brought severe discomfort and extreme inflammation and I was told that it wouldn't get better without a major medical treatment. After finding out about Cerule and taking StemEnhance Ultra twice a day, I am back playing soccer even better than before and with ease! I am so very grateful for this product, each and every day."
Jeanne Venturino, ID
"I was introduced to StemEnhance Ultra about 2 1/2 years ago. Although I felt very healthy, as an RN, I understood the wellness concept and knew the needs of the aging body. Very shortly after starting on the product, I realized I wasn't having the many severe headaches I had dealt with month after month, for many years. In just a few months, I was feeling better than I've felt in years! I realize what a blessing Cerule has meant to my life and overall feeling of well being."
 Alexander L, NC, Boxing
Alexander L, NC, Boxing"I'm a full-time student, varsity athlete and I work part-time, so I have long demanding days and very short nights.
StemEnhance Ultra gives me the power and energy I need to excel at everything."
Jovi Olsen, Washington State
"I injured my ankle and foot and it just wouldn't heal not matter what pyhsical therapy or other strategies I tried. Then I started taking StemEnhance Ultra and PlasmaFlo and within six or seven weeks it stopped hurting!"
 Allan Ahrens
Allan Ahrens"I injured my arm last October.
With StemEnhance Ultra and physical therapy, I am seeing the mobility of my arm returning, and strength is building up in the muscles that had atrophied from disuse.
StemEnhance Ultra is helping all those millions of adult stem cells to do their powerful work to bring my arm back!"
 Joan H, Fitness Enthusiast
Joan H, Fitness Enthusiast"I run nearly two miles on the treadmill in 30 minutes!
Not bad for a 62 year old who hadn't been on any type of exercise program for a long time!
StemEnhance Ultra has really given me a great start and now I am sure I will reach the desired health and energy I have always wanted.
Thank you Cerule!"
 Peggy Z
Peggy Z"I had serious eye issues and had to have immediate surgery for each eye.
I was told I would never read again.
That's all gone away since I've been on StemEnhance Ultra."
 Vincent H
Vincent H"I never expected my hair to grow like this.
This is the result of 3 months using StemEnhance Ultra."
 Marjorie LS
Marjorie LS"After including StemEnhance Ultra in my diet, my vision is improving.
It sounds impossible, but after five weeks, I can definitely recognize a difference."
 DeWayne F
DeWayne F"After 2 weeks on StemEnhance Ultra, my blood sugar stabilized in the normal range, and all discomfort in my legs and feet disappeared."
 Nancy E
Nancy E"Since taking StemEnhance Ultra, my skin condition has practically disappeared.
I have no pain and take no medications for pain.
My knee surgery has been cancelled."
 Haripriya D
Haripriya D"After about 10 days on StemEnhance Ultra, the pain in the knee was almost gone and I could walk without my cane.
A very slight sensitivity still exists, but it seems to continue to improve as time goes on."
Matthew Crown from Montana
"I have an ongoing hip condition that exercise, chiropractors, and physical therapy have helped somewhat but I seemed to have reached a plateau. StemEnhance Ultra has brought tremendous differences in just a few weeks. It seems like the brain/body connection is much stronger. Day by day I walk with more and more confidence, instead of the stiffness and timidity of an older man and it feels GREAT!"
100% Satisfaction Guarantee
 Our Guarantee is simple: If you're not satisfied with our products for any reason, please contact us immediately and we will either provide a replacement, or a refund, your choice.
Our Guarantee is simple: If you're not satisfied with our products for any reason, please contact us immediately and we will either provide a replacement, or a refund, your choice.If requesting a refund, simply contact us within 30 days of purchase. We are willing to work with you to ensure your satisfaction!

StemEnhance Ultra Frequently Asked Questions
What is StemEnhance Ultra?StemEnhance Ultra concentrates and combines extracts from nature's most primitive superfoods, fresh water microalgae and marine macroalgae, providing the body with stem cell nutrition and the ultimate in stem cell support.
Why is StemEnhance Ultra beneficial?
StemEnhance Ultra assists the body's inherent ability for long-term self-renewal by supporting the body's natural release of bone marrow stem cells.
What are StemEnhance Ultra side effects?
The only side effect of taking StemEnhance Ultra is that you feel great. The body is in a constant conflict between break down and renewal. You simply feel better when the balance is tipped towards renewal.
What are the ingredients in StemEnhance Ultra?
StemEnhance Ultra provides the ultimate in stem cell support containing a proprietary blend of highly concentrated extracts, including Fucoidan, Cerule's own exclusive patented ingredients, StemEnhance (Aphanizomenon Flos-Aquae concentrate) and Mesenkine.
StemEnhance Ultra is shown to support the release of stem cells from the bone marrow.
Fucoidan (Undaria Pinnatifida) is a marine algae well known to support the immune system. Cerule's fucoidan comes from undaria harvested from pristine environments like the Tasman Sea and Patagonia. Fucoidan from undaria pinnatifida has been documented to increase the number of circulating stem cells.
Mesenkine is a unique extract from Spirulina, isolated through our patented extraction process, that supports the release and homing of stem cells by balancing key messengers involved in stem cell function.
StemEnhance Ultra does not contain dairy, wheat, gluten, peanut, soy, corn, or allergens. There are no artificial flavors or colors. It is 100% vegetarian, non-GMO, and free from herbicides and pesticides.
What are stem cells and why are they considered important?
'The primary roles of adult stem cells in a living organism are to maintain and repair the tissue in which they are found.' http://stemcells.nih.gov/info/basics/4.htm. Furthermore, stem cells released from the bone marrow can migrate to various tissues where they contribute to the process of tissue repair.
How do I take StemEnhance Ultra?
Suggested usage is 2 capsules 1 to 2 times daily. Others have experienced great results taking 4 capsules 1 to 2 times daily.
What if I can’t swallow capsules?
The capsules can be opened, and the contents can be mixed with food. A pureed texture is recommended.
Can children consume StemEnhance Ultra?
The clinical studies were done using adults therefore we recommend StemEnhance Ultra for adult consumption, however there are no known contraindications for children.
Can pets consume StemEnhance Ultra?
StemEnhance Ultra is formulated for human consumption. We know of no reason that it may be harmful to pets. AFA and spirulina have been used in the pet nutrition industry for years. However, no studies have been done using the product for pet consumption. Please consult with your Veterinarian.
Is there research on StemEnhance Ultra?
Through multiple clinical trials, StemEnhance Ultra was documented to optimize stem cell function in the body by increasing the number of both stem cells and and Endothelial Progenitor Cells (EPCs) in the bloodstream, supporting optimum renewal and repair of tissues and organs.
StemEnhance Ultra also contains Mesenkine, that was shown to increase the blood concentration of G-CSF that plays a key role in stem cell release.
What is the StemEnhance Ultra capsule made from?
As stated on the label, the vegetarian capsule is made from hypromellose. Hypromellose is cellulose derivative or plant fiber.
Is StemEnhance Ultra certified Kosher and Halal?
StemEnhance Ultra ingredients are certified Kosher.
It is not certified Halal.
What is the shelf-life of StemEnhance Ultra?
There is an expiration date at the bottom of each bottle. StemEnhance Ultra has a shelf life of 3 years from manufacturing. All bottles should be stored in a cool, dry place.
Can StemEnhance Ultra be consumed with other Cerule products and other nutrients?
Yes, the Cerule products can be consumed together and were designed to enhance the beneficial effects of each other. We know of no concerning interaction between the Cerule products and other nutritional supplements.
Are there any known nutrient-medication interactions?
Like many green foods, StemEnhance Ultra contains naturally occurring vitamin K, which could interfere with vitamin K blockers used to thin the blood.
If you have any health condition and/or are using medication, then consult your attending health care provider before consuming any nutritional supplement.
For some people, due to their conditions and medications, they need to manage their intake of certain nutrients. Below are the amounts of naturally occurring nutrients found in the plant based ingredients within StemEnhance Ultra:
Vitamin K: around 20 ug per serving (2 capsules)
Iron: 0.34 mg per serving (2 capsules)
Iodine: around 4 ug per serving (2 capsules)
Sodium: 9.66 mg per serving (2 capsules)
PEA: >0.5%
Why does the StemEnhance Ultra label state 'consult your physician if you are pregnant or nursing?'
Pregnancy and nursing are considered special conditions. We recommend that your attending Doctor(s) be made aware of any and all supplements consumed during this time. At this time, we do not advise StemEnhance Ultra consumption during pregnancy.

Distributed by: Cerule LLC
Irvine, CA 92612 USA





